This content is also available in:
Italiano
Português
Čeština
Română
Türkçe
Figures 9d-1 to 4 in this chapter are taken from Chapter 2 in the European guidelines for quality assurance in cervical cancer screening: http://screening.iarc.fr/doc/ND7007117ENC_002.pdf).
Central register
In an organised programme a central register should hold patient details (name. date of birth, address), registered GP name and address, vaccination status, screening history (previous tests and coded results) and links to laboratory results (cytology and histology), colposcopy findings and the national cancer registry.
Information to women
Information leaflets
- Information leaflets should be provided to women before the cytology or HPV test is performed explaining the procedures for repeat tests or investigation of abnormal results.
- Contact details for personal advice should be included in the leaflets
- Information leaflets explaining the benefits and limitations of screening should be provided before woman consent to have their cytology or HPV test verbally or in writing as required by local or national protocols.
Result letters
- Women should be informed in writing of their cytology results in a timely manner with an explanation of what the HPV results, terms and cytology categories mean.
- Contact details should be given for further information and personal consultation at the clinic they attended for the test or their GP.
Invitation letters
Invitation letters should be sent to women according to local or national protocols for age range and intervals for routine screening (e.g. call at age 24, 3-yearly to 49, 5-yearly to 64 as in the UK or variations as required in different countries). The system in Denmark is one example of a call and recall system, which takes note of women who do not want to take part in the programme.
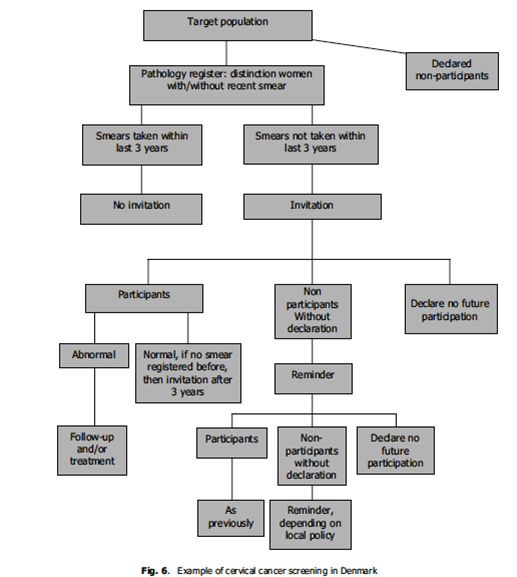
Invitations for screening
|
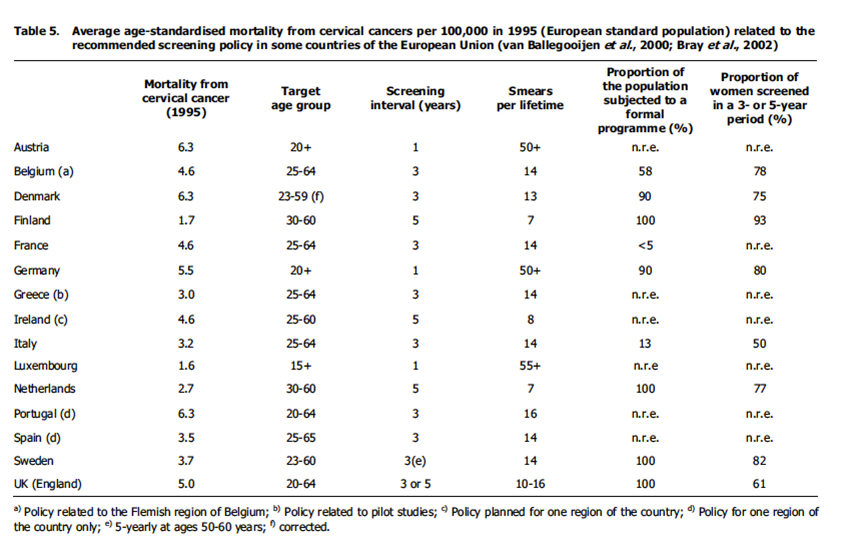
Intervals for screening and age groups screened
Intervals and age ranges vary throughout Europe and worldwide: invitations from age 20-25 years to 64 years at 3-5 yearly intervals are the general rule as shown in Figure 9c-2.
Cytology results
Cytology results should include a text report, a classification translatable into the Bethesda system (Inadequate, with reasons; NIMLS; ASC-US, ASC-H, AGC, LSIL, HSIL, cancer) and a recommendation for further action.
HPV tests
HPV tests should be carried out in an accredited laboratory and should be requested by the cytology laboratory according to the cytology test result and local protocols for triage and test of cure.
HPV results should be included in the cytology report
Recommended action
Depends on local and national protocols for screening intervals, HPV triage etc.
Routine recall
- Women with negative results and negative screening histories should be recalled after 3 5 years depending on regional and national protocols.
Early recall (3, 6 or 12 months as agreed in regional or national programme)
- Inadequate tests should be repeated in 3 months since immediate repeat tests may be unreliable.
- First occurrence of ASC-US and LSIL may be repeated in 3 or 6 months depending on regional or national protocols for referral and availability of HPV triage
- Negative cytology as follow up of previous ASC-US or LSIL that was managed by cytological surveillance.
- Treatment of CIN: duration of follow up depends on regional or national protocols and availability of HPV test of cure.
- Positive HPV with negative cytology depending on age, screening history and regional and local protocols.
Recommendation for colposcopy
HSIL, ASC-H, AGC, AIS or cancer in asymptomatic women.
LSIL (first or second occurrence depending on regional or national protocols)
ASC-US / HPV-positive
Negative cytology / HPV-positive (first or second occurrence depending on regional or national protocols and age of patient)
Recommendation for gynaecological referral
Symptoms that might be related to invasive cancer (e.g. post-coital bleeding, post-menopausal bleeding) whatever the result of the cytology test
Abnormal cytology suggesting cervical cancer in symptomatic women
Cytological changes suggesting non-cervical neoplasia
Reminders to women
Reminders should be sent from the central register, laboratory or colposcopy clinic depending on regional or national protocols to remind women for repeat tests or appointments, with defaulter letters for non-responders
Details of reminders and responses should be held on screening records.
Regional and national variation
|
The European guidelines for quality assurance in cervical cancer screening as quoted in this chapter are available online: http://screening.iarc.fr/doc/ND7007117ENC_002.pdf
NHS Cervical Screening Programme guidelines are available online: http://www.cancerscreening.nhs.uk/
The American Society for Colposcopy and Cervical Pathology are available as: www.asccp.org/Portals/9/docs/ASCCP%20Management%20Guidelines_August%202014.pdf

