It is good practice to know the variation in appearances of HSIL.
- Keratinizing
- Non-keratinizing large cell
- HSIL in immature squamous metaplasia
- Small cell severe dyskaryosis
- HSIL in atrophic samples
Keratinising
Presentation
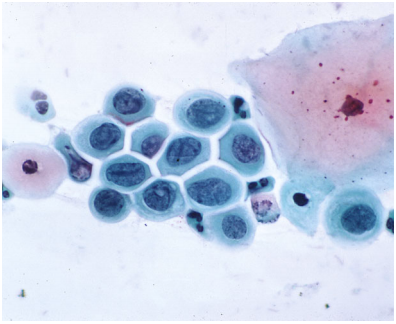
There may be an abundance of material on the cytology preparation due to the mature nature of this dysplasia. Cells appear as sheets with crowded nuclei, overlapping nuclei with single cells dissociating from the groups. In LBC preparations dissociation is more evident due to the mechanical nature of the preparation.
Tumour diathesis is absent even though at times the cells may suggest invasive carcinoma, which is an important differential diagnosis: as well as tumour diathesis, haemorrhage and tissue fragments should be looked for.
On the other hand, HPV infection may be suggested by parakeratosis and dyskaryosis making this type of SIL difficult to grade (Faquin et al. 2001); the NC ratio may not be as high as expected in HSIL.
Nucleus
The nuclei of keratinising HSIL are often pyknotic and hyperchromatic, on the verge of being black. Some of the cells will have a high NC ratio but in others it may be lower than expected for HSIL. If the chromatin is visible it will be coarse and irregularly distributed allowing the diagnosis of HSIL to be made.
Cytoplasm
As the lesion is keratinising, the cytoplasm will be dense and refractile. The cytoplasm is generally eosinophilic or orangeophilic but may sometimes be cyanophilic.
Figure 9c-12 (a, b). HSIL favour CIN3 (high-grade dyskaryosis, severe) with keratinisation confirmed to be CIN3
[FIGURES]
Non-keratinizing large cell HSIL
Presentation
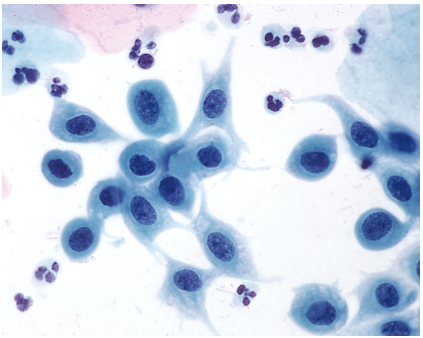
There is generally an abundance of material on the cytology preparation. The abnormal cells will present in groups, sheets and single cells. The groups and sheets may be crowded and architecturally disordered as the cells lose their polarity. Single cells will be dissociated from the groups and sheets.
Nucleus
The nuclei will show variation in size and shape but the shape is generally round to oval. Moulding of nuclei may be a feature in this form of HSIL. Irregular nuclear borders can be appreciated in the dissociated single cells. Although they should never be used to make a definitive diagnosis, bare abnormal nuclei are a feature.
The chromatin, in this case, is variable as there are variations in presentation.
- Nuclei may be hypochromatic with bland chromatin
- Nuclei may be normochromatic with a finely granular but irregular chromatin
- Nuclei may be hyperchromatic with granular, coarse, irregular chromatin.
Cytoplasm
The cytoplasm is delicate (and there may be bare nuclei) with indistinct cell borders. It may be difficult at times to decide if the cells are squamous or glandular in origin. The cytoplasm is basophilic or almost colourless.
Nuclear/cytoplasmic ratio
In view of the poorly defined cytoplasmic margins and delicate cytoplasm NC ratio may be difficult to assess. The diagnosis is made on the abnormal chromatin pattern and irregular nuclear membranes (dyskaryosis) and the disorderly arrangement of cells.
Figure 9c-13
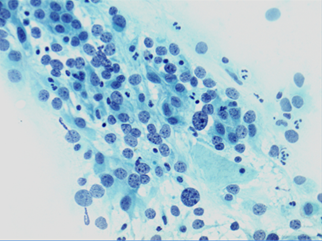
HSIL in immature squamous metaplasia
HSIL typically involves the TZ of the cervix, which is derived from immature metaplasia. Thus it may be difficult to interpret when the metaplasia remains immature (see also ASC-H below).
Presentation
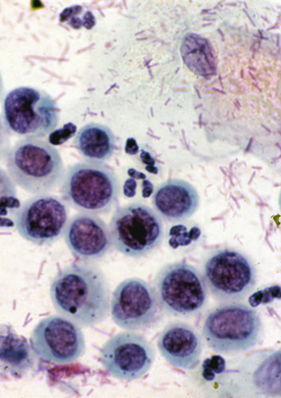
These cells usually present as single cells as opposed to immature metaplastic cells, which are usually in small sheets. A very important feature of these cells is that on conventional cytology they can be seen trapped in streaks of mucus, which at times can be helpful in searching for more abnormal cells. The cells vary in size and shape, a feature of their abnormal nature. This lesion can be seen co-existing with other HSIL appearances.
Nucleus
The NC ratio of a normal immature squamous cell is higher than that of an intermediate cell and may approach 50%. The diagnosis of HSIL depends on the abnormal chromatin pattern and nuclear membranes. The nuclear membranes are irregular and may be indented; the nuclei are pleomorphic with marked size variation between the abnormal cells; the chromatin is usually hyperchromatic and coarsely granular.
Cytoplasm
The cytoplasm is thick, but not refractile, with well-defined cell borders. Vacuoles may be present. The NC ratio must be considered in comparison with that of normal immature metaplastic cells but is likely to be more than 50% in HSIL
Figure 9c-14 (a-b). HSIL in immature metaplasia
HSIL in immature metaplasla showing the abnormal chromatin pattern, attenuated cytoplasm and higher NC ratio than seen in
Small cell severe dyskaryosis
Presentation
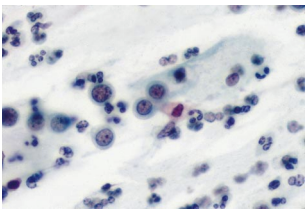
This is an uncommon well-recognised presentation of HSIL in CIN3 although it should be remembered that nuclei in HSIL are usually smaller than LSIL. In small cell severe dyskaryosis the nuclei may be little larger than lymphocytes, polymorphs or normal intermediate squamous cell nuclei. There is generally abundant material on the cytology preparation and the abnormal cells with be either in hyperchromatic crowed groups or single cells. The groups may be crowded with no definition to the cytoplasmic cell borders. The nuclei will show a loss of polarity. This type of HSIL is seen in CIN3 rather than CIN2 and is hardly ever combined with LSIL or CIN2.
Small cell severe dyskaryosis may show signs of neuroendocrine differentiation in which case the cells will be larger, oval rather than round and may show nuclear moulding in cohesive cell groups.
Nucleus
The nuclei are usually round and of similar size and shape in each case. They are usually hyperchromatic with a relatively uniform distribution of chromatin. The chromatin varies from fine to coarse and may be pale rather than hyperchromatic as explained by Smith and Turnbull (1997). Nuclear membrane irregularities and indentations are frequently seen.
Cytoplasm
Due to lack of maturation of the NC ratio may approach 100% and will be higher than immature metaplastic or parabasal cells. The cytoplasm may be inconspicuous; when visible, it will be basophilic.
Differential diagnosis
The differential diagnosis of small cell dyskaryosis consists of non-malignant entities such as follicular cervicitis, endometrial cells and immature squamous metaplasia. Once recognised as HSIL it is likely to be reported as high-grade dyskaryosis (severe) and cannot be confused with LSIL.
The differential diagnosis includes small cell neuroendocrine carcinoma, which may be suggested by the larger cell size, coarse granularity and moulding of oval nuclei, and the presence of tumour diathesis, blood and frequent mitoses.
Figure 9c-15. Small cell severe dyskaryosis (HSIL favour CIN3).
(a) Nuclei are little larger than those of intermediate cells or neutrophil polymorphs; the NC ratio is well over 50% (x100 magnification).
HSIL in post-menopausal samples
Atrophic samples are innately difficult as the atrophic pattern is composed of hyperchromatic parabasal sheets of cells with a high NC ratio in a background of inflammation and autolysis. All these features are seen in HSIL.
Attention should be paid to samples with a separate population of cells, which should stand out from the uniform atrophic pattern. HSIL will show the features of dyskaryosis: irregular chromatin, anisocytosis and irregular nuclear membranes. In the presence of atrophic vaginitis, when the normal cells may be enlarged and somewhat atypical, a short course of topical oestrogen should be recommended. HPV triage may be useful in this situation.
Figure 9c-15 (a-b).