This content is also available in:
Italiano
Português
Deutsch
Čeština
Română
Türkçe
The cervical smear test was developed by Dr George Papanicolaou, an American anatomist who was born and studied in Greece. He was engaged in research into the effect of ovarian hormones on the epithelium of the female genital tract.
In 1926, Dr Papanicolaou reported that tumour cells could be seen in vaginal secretions of women with cervical cancer, leading to his publication Diagnosis of uterine cancer by the vaginal smear in 1943. The first cervical cancer screening clinic was opened in Massachusetts in 1945.

Cytology as a screening test the Pap smear
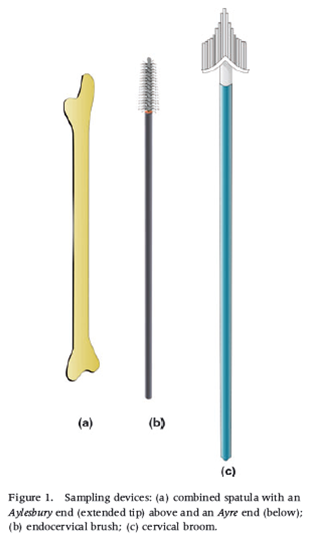
The cervical smear, or Pap test, was developed as a simple, cost-effective and sensitive test that has been used worldwide as a screening test for more than 50 years. In the UK alone, over 3 million cervical smears are performed annually. The test involves gently scraping a sample of epithelial cells from the surface of the cervix using a spatula, brush or other sampling device.
In conventional cytology the cells are directly transferred from the spatula or brush to the slide and then fixed using a spray fixative or alcohol. These days, we often use liquid-based cytology as the preferred method of sample collection as this enables the laboratory to produce its own slides as well as having left over tissue for axillary tests. Whichever method is used, the end product is that cells are thinly spread on a slide for microscopic evaluation.
Cytological screening
The initial microscopy is carried out by a cytotechnologist (primary screener) who is trained to detect abnormal cells amongst the many thousands of normal cells in the smear or LBC preparation. The samples that contain abnormal cells are passed on to a senior scientist or consultant pathologist who grades the abnormality according to a recognised terminological system and provides a clinical recommendation to the clinicians on how to recommend management of the patient.
This simple and cost-effective test has saved countless lives since its introduction through the treatment of pre-invasive lesions.
The Pap test as part of a national screening programme
The primary aim of the cervical smear test is to prevent the development of invasive cancer of the cervix by detecting pre-cancerous lesions known as cervical intraepithelial neoplasia (CIN) in the uterine cervix, which is usually asymptomatic and only found by screening. Conservative treatment of CIN by local ablation or excision of the abnormal epithelium significantly reduces the risk of invasive disease.
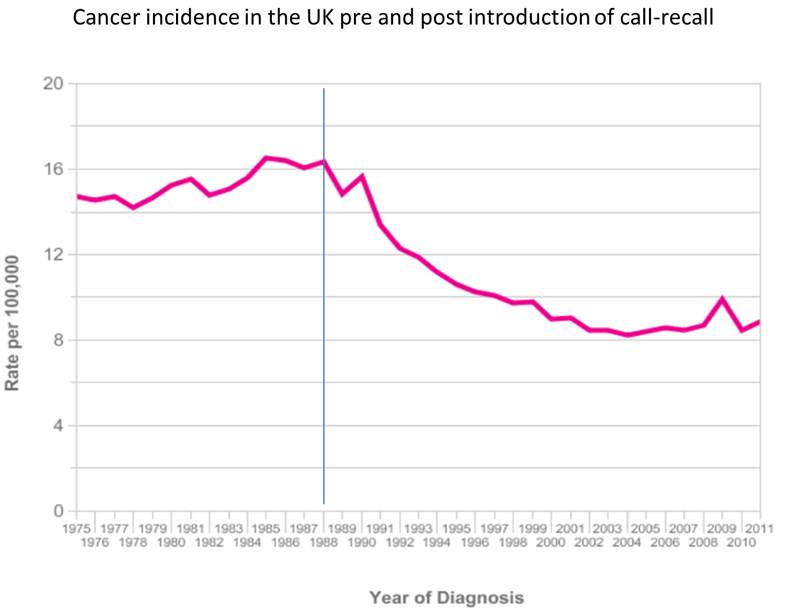
Occult cancer, before a tumour is visible and usually at an early and treatable stage, may also be detected by cytological screening.
Although cytology tests are sometimes carried out during investigation of women who are symptomatic or have clinically evident cancer, such tests may be unreliable, There is an increased risk of the cytology sample being reported as negative or inadequate, due to contamination by blood, inflammatory cells and necrotic debris associated with invasive cancer, obscuring the abnormal cells in the smear. [Link to Chapter 8 Collecting and processing cellular samples from the cervix]
Women with symptoms or signs suggesting invasive cancer should be referred for gynaecological investigation whatever the result of a cytology test.
The role of human papillomavirus (HPV)
The discovery of HPV as the main aetiological agent in cervical cancer and its precursors has revolutionised cervical screening and the role of cytology in the screening process.
HPV testing is now recommended for triage of women with atypical/borderline cytology and during follow-up after treatment of CIN (test of cure). Although cytology is a sensitive method of detecting recurrence, women who have been treated for high-grade CIN are at increased risk for cancer and HPV testing is more sensitive than cytology alone for detecting recurrence.
In many countries primary HPV screening is being introduced because it has been found to be more sensitive than cytology for detection of high-grade CIN although its specificity is substantially lower. This fundamentally changes the role of cytology from a screening to a diagnostic test. However, it does not change the principles of cervical cytology or the vital importance of quality control.
Treatment of precancerous lesions
In the early days of screening treatment was confined to carcinoma in situ, now referred to as cervical intraepithelial neoplasia grade 3 (CIN3). Knife cone biopsy and hysterectomy were the only treatments available.
Ablation or excision of the CIN lesion and the transformation zone was developed during the 1990s as a more conservative but highly effective method of treatment: large loop excision of the transformation zone (LLETZ). LLETZ is now the method of choice although knife cone biopsy, trachelectomy and even hysterectomy may be carried out for widespread lesions, glandular neoplasia and occult invasive cancer.
Avoiding risks of treatment, which are related to the depth of excision biopsy, represent an important consideration in cervical screening.
HPV vaccination
The development and introduction of HPV vaccination, which was approved as a vaccine in 2005, will further revolutionise the practice of cervical cytology, colposcopy and treatment of cervical cancer precursors. Vaccinated women are already entering the screening age groups in countries such as Australia and the expected effect of reducing the prevalence of genital warts and CIN are being recorded.
As a result of the falling prevalence of CIN and cancer, the accuracy of cytological screening sensitivity and PPV may be compromised, which is one of the reasons primary HPV testing is being introduced in many countries. Nevertheless, the low specificity of HPV testing requires cytology as a diagnostic triage test highlighting again the vital importance of quality control.
Learning points from Chapter 1
|

