This content is also available in:
Português
The ability to screen a predominantly negative cytology slide and notice occasional abnormalities among many thousands of cells requires specialist training, knowledge of the nature of normal and abnormal cells, and concentration and dedication. Here we deal with sources of error in screening and how they can be avoided.
Sources of error
Personal reasons
- Habituation, which is a subconscious process by which the brain ignores or switches off because of a repeated signal.
- Distraction by telephone calls, other interruptions and thoughts about unrelated personal concerns may lead to loss of concentration.
- Over-confidence, experience that an abnormal slide can often be recognised from the first field, may lead to occasional abnormal cells being missed.
- Misunderstanding of subtle cellular changes not recognised during training may result in experienced screeners continuing to make the same mistakes.
- Screening too many slides during the same day may lead to loss of concentration
- Hurrying through work at the end of a long day may lead to abnormalities being missed.
- Anxiety about missing abnormalities may lead to over-calling benign cell changes as atypical or borderline.
Environment and equipment
- Unsatisfactory microscope optics and ergonomics may lead to personal discomfort that may affect performance.
- Screening areas not protected from other laboratory activities
- Insufficient space for slides, computers, notepads etc.
Known causes of false negatives
- Small, pale and sparse cells are at risk for being missed on primary screening with conventional or liquid-based cytology (Demay 2000; Gupta et al. 2013; Leung et al. 2008; Mitchell & Medley1995).
- Hyperchromatic crowded cell groups may be overlooked and thought to be endocervical or endometrial cells and liquid-based cytology (Demay 2000; Gupta et al. 2013; Robertson & Woodend 1993).
- Glandular abnormalities misinterpreted as reactive endocervical cells
- Low prevalence of abnormal cells in the preparations may reduce sensitivity of screening (Evans et al. 2011).
Known causes of false positives and false negatives
Reactive and metaplastic changes thought to be dyskaryosis.
Endometrial cells, histiocytes or lymphocytes misinterpreted as neoplastic.
Sources of error in primary screening
|
Methods for quality control and quality assurance
Training and regular update of cytology screeners should be mandatory for all grades of cytotechnologists (QC).
Rescreening of slides before the final report is issued is a useful method of internal quality control method that is recommended in the European guidelines and has been shown to be more accurate than 10% full rescreening of slides (Wiener et al. 2007; Arbyn et al. 2003). There are several methods:
Rapid rescreening
Rapid rescreening has been demonstrated to be an effective method of reducing false negative rates thought to be due to fatigue or misinterpretation (Faraker & Boxer 1996)
- Each slide is partially reviewed for 1 minute at x10 magnification and abnormalities are passed on to a senior cytotechnologist or pathologist for reporting.
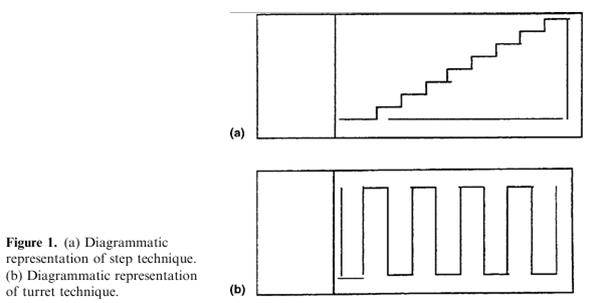
- Stepwise screening was found by Dudding et al. (2001) to be more effective than whole slide/random or turret screening (Figure 11.1). Accuracy varied between individuals and declined after more than 50 slides had been examined.
- Rapid rescreening has been shown to be more effective in reducing false negative rates than fully rescreening 10% of slides (Manrique et al. 2006). Rapid screening of all negative and inadequate slides is the recommended method of internal quality control in the UK, either as rescreening or pre-screening (NHSCSP 2013).
Rapid pre-screening
Rapid pre-screening of all slides before they are subjected to routine screening (abnormal cells being recorded but not marked on the slide) has been found to be equally sensitive to rapid rescreening (Arbyn et al. 2003).
Pre-screening has the advantage of allowing the rescreening process itself to be monitored and its sensitivity (against the final result) to be compared with full primary screening.
Rapid pre-screening is more interesting and rewarding than rescreening negative slides because there are more abnormal cells to be looked for, which may have a positive effect on sensitivity (Evans et al. 2011).
Pre-screening has been shown to be more sensitive in LBC than conventional smears (Dudding et al. 2011a) and its sensitivity may improve with time (Dudding et al. 2011b).
Automated screening
Automated screening is expensive but may provide an even more accurate method of quality control if used as a pre-screening method followed by full primary screening (Heard et al. 2013). Unlike rapid rescreening or rapid pre-screening, screening with an automated system is at least as sensitive as full primary screening (Roberts et al. 2007) and has been accredited for that purpose.
Assessing personal and laboratory performance of primary screening
Rapid review, rapid pre-screening and 10% full rescreening all provide a method for internal and external quality assurance of personal and laboratory performance of primary screening, which is the critical stage of the process whether cytology is used as the primary screening test or as triage after primary HPV screening.
Sensitivity of primary screeningNumber screened as abnormal x 100
___________________________________________________________
Number of abnormal final reports (ASC-US+ or HSIL+) |
All these methods of rescreening or pre-screening detect abnormalities before the final reports are issued but very few additional abnormalities are found: 0.18% (CI 0.14-0.21%) for rapid rescreening and 0.19% (CI 0.03-0.35%) for rapid pre-screening (Arbyn et al. 2003). This slight increase in sensitivity provides a powerful tool for assessing performance of cytology screeners but should be taken in the context of the following additional factors.
- Managing screening errors should be confidential and sensitive bearing in mind that the most competent screener will miss occasional high-grade abnormalities.
- Remedial action, such as additional training or update, should only used when a pattern of poor performance is found.
- Since the final report depends on accuracy of that of the pathologist sensitivity of screening should be taken in the context of individual variations in reporting rates in the laboratory.
- Peripheral reasons for poor performance, such as excessive workloads, poor equipment and unsatisfactory laboratory conditions should be dealt with accordingly.
QC and QA of primary screening
|

