This content is also available in:
Italiano
Español
Čeština
Magyar
Polski
Definition of Quality Assurance, Quality Control & Quality Standards
Quality Assurance= The process of building quality control measures into a system which will ensure a consistently high standard of performance.
Quality Control = The set of measures designed to ensure the accuracy of interpretation and reporting of cervical smears.
Quality Standards= Measurements of laboratory performance agreed at local, national or international levels which reflect good cytology practice.
Types of quality assurance
Quality Assurance systems may be internal or external
- Internal Quality Assurance systems (IQA) are applied by the laboratory on a daily basis such as in regular checks on data entry and introduction of tiered screening.
- External Quality Assurance systems (EQA) are applied by an outside regulatory body e.g. proficiency testing schemes, accreditation.
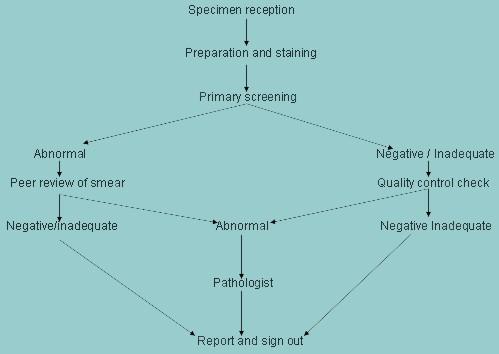
Internal quality assurance (IQA) measures to be taken on a daily basis
| Errors encountered | IQA measures |
|---|---|
| Specimen reception | |
| Errors at the point of reception are usually due to mismatching of smears and request forms. |
|
| Specimen processing | |
| Poor quality staining and fixation is the single most likely cause of errors of reporting cervical smears. |
|
| Specimen interpretation | |
| The commonest error is false negative reporting |
|
The analysis of cervical smears is a skilled repetitive task requiring intense concentration by the screener who is required to detect a relatively small number of abnormal cells among the many thousands of normal epithelial cells in the smear. Consequently the risk of false negative reporting is high. Quality assurance measures to reduce the risk of false negative reports must be in place every cytology laboratory.

