This content is also available in:
Italiano
Español
Comparison of adequate smears against inadequate smears:
| Satisfactory for evaluation | Unsatisfactory for evaluation |
|---|---|
| Appropriate labeling | Lack of patient data |
| Well displayed and well preserved squamous epithelial cells covering more than 10% of the area under the cover slip. | Scant squamous epithelial cells component : less than 10% of the slide surface |
| Presence of endocervical and or immature metaplastic cells |
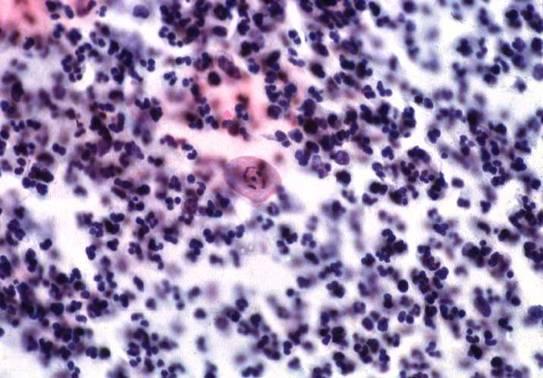
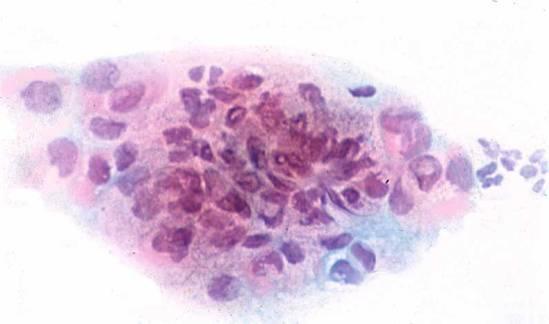
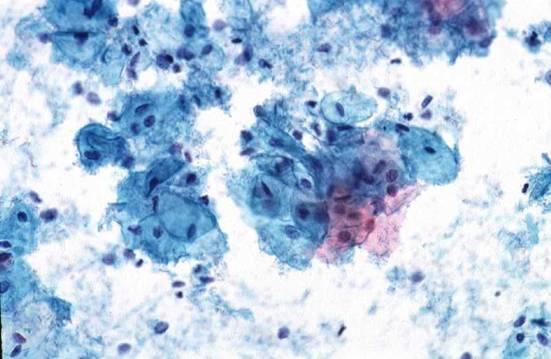
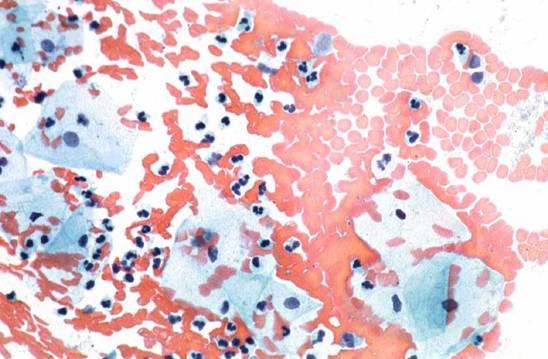
Obscuring blood, inflammatory cells, excessive cytolysis, thick areas, poor fixation, air drying |
| Clinical information provided age, menstrual status, appearance of cervix , irregular bleeding e.g. post coital or post menopausal bleeding. | Contaminants which obscure more than 50% of epithelial cells |
A SLIDE CONTAINING ABNORMAL CELLS SHOULD NEVER BE CLASSIFIED AS INADEQUATE.
Here are some examples of inadequate smears:
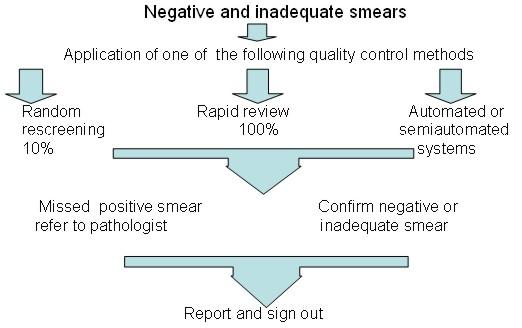
Quality control measures to reduce the risk of false negative smears
- Random re-screening: This method is widely used in the United States. It involves supervisory staff re-screening 10% of all smears classified as negative or inadequate by the primary screener. The whole slide should be examined allowing approximately 6 minutes per smear. Although the chances of detecting error are small, this approach increases awareness of risk of error.
- Rapid re-screening: This method is widely used in the UK. It involves supervisory staff performing a partial review of every slide classified as negative or inadequate by the primary screener. The slide is reviewed for 1 minute at low magnification (x10 objective) A record of the relative sensitivity of primary screening should be kept using the following formula: [Abnormal smears correctly identified by the screener / total abnormal smears after rapid review x 100]
Internal quality control procedures which should be applied periodically
- Peer review and regular discussion of abnormal smears
- Comparison of biopsy with cytology
- Review smear history of women with invasive cancer for possible false negative smear reports Slides should be stored for up to 10 years for this purpose
- Check on Workload and Monitoring turnaround time.
- Statistical monitoring of laboratory performance at regional or national level.

